Erenumab is one of the most widely recognized CGRP-targeting therapies used for migraine prevention in adults. Approved under the brand name Aimovig, the medication represents an important shift in preventive migraine treatment, particularly for patients who do not respond adequately to traditional therapies.
Unlike older migraine medications originally developed for epilepsy, hypertension, or depression, erenumab was specifically designed to target migraine-related biological pathways. The therapy works by blocking the calcitonin gene-related peptide (CGRP) receptor, which plays a central role in migraine pathophysiology and pain signaling.
As migraine management increasingly moves toward targeted neurological care, biologic therapies such as erenumab are becoming more important within preventive treatment strategies. Neurologists and headache specialists are also paying closer attention to long-term efficacy, patient quality of life, treatment adherence, and safety outcomes associated with CGRP inhibitor therapies.
Erenumab reflects a broader transition in migraine care from generalized symptom management toward targeted biologic intervention focused on migraine-specific mechanisms.
Erenumab Uses
It is primarily used for the prevention of episodic and chronic migraine in adults. The therapy is commonly prescribed for patients experiencing frequent migraine attacks, migraine with aura, or inadequate response to conventional preventive medications.
Unlike acute migraine therapies used during attacks, functions as a preventive treatment intended to reduce monthly migraine days, migraine severity, and migraine-related disability. Healthcare providers often consider erenumab when patients continue experiencing significant migraine burden despite trying older preventive therapies.
Aimovig: Brand Name, Manufacturer & Formulations
Aimovig is manufactured by Amgen in collaboration with Novartis. The medication is available as 70 mg and 140 mg prefilled SureClick autoinjector formulations designed for subcutaneous self-administration.
At present, there is no approved erenumab tablet formulation available.
Erenumab Dosage & Administration
| Dose | Indication | Administration | Adjustment Criteria |
| 70 mg monthly | Episodic migraine prevention | Subcutaneous injection | Standard starting dose |
| 140 mg monthly | Chronic or refractory migraine | Subcutaneous injection | Considered for inadequate response |
Its injections are typically administered once monthly into the abdomen, thigh, or upper arm. Patients are generally advised to rotate injection sites and allow the medication to reach room temperature before administration.
Healthcare providers may adjust dosing based on migraine frequency, treatment response, tolerability, and previous preventive therapy history.
How Erenumab Works?
Development, Pharmacokinetics and Pharmacodynamics of Erenumab
Erenumab was developed as a fully human monoclonal antibody targeting the CGRP receptor pathway associated with migraine generation and neurovascular signaling. Following subcutaneous administration, the drug maintains prolonged receptor activity due to its biologic half-life, allowing monthly dosing schedules that may improve treatment adherence.
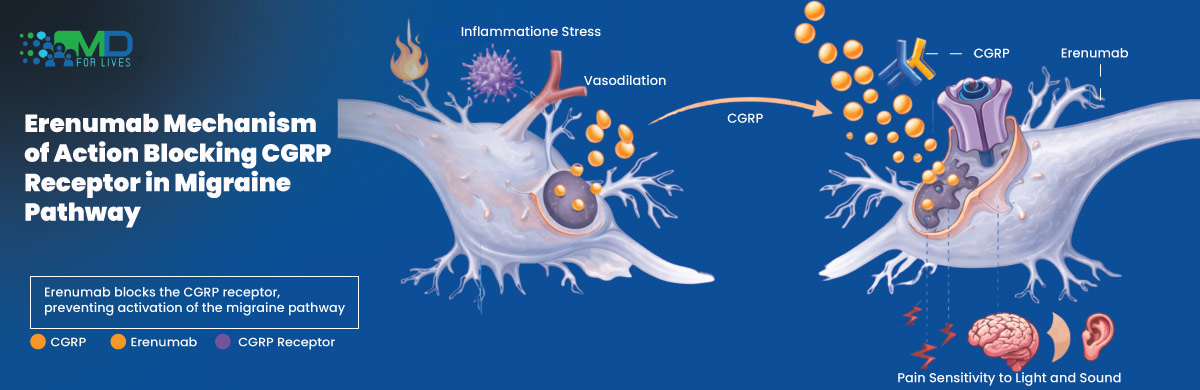
Erenumab Mechanism of Action
Erenumab works by blocking the calcitonin gene-related peptide (CGRP) receptor involved in migraine signaling pathways.
CGRP is a neuropeptide associated with vasodilation, neurogenic inflammation, and pain transmission during migraine attacks. By inhibiting the receptor itself, erenumab helps reduce migraine frequency and severity in susceptible patients.
This mechanism differs from traditional migraine preventive therapies that indirectly influence neurological activity without specifically targeting migraine biology.
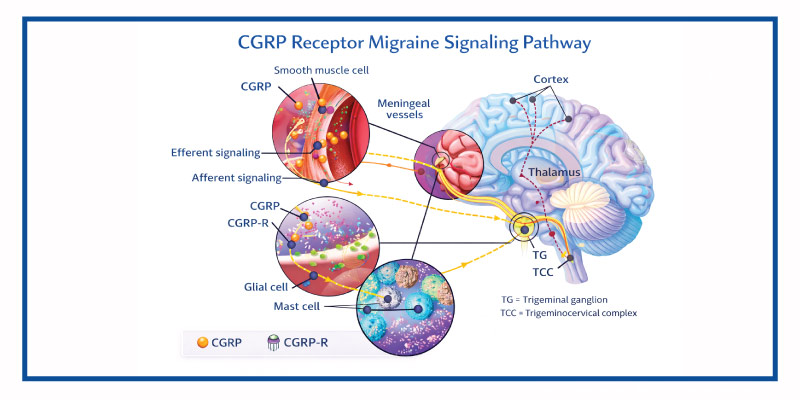
Role of Erenumab in the Treatment of Migraine
Migraine management has historically relied on medications originally developed for cardiovascular or neurological conditions. Although many patients benefit from these therapies, tolerability and inconsistent response remain important clinical challenges.
The introduction of CGRP inhibitors changed preventive migraine treatment by providing therapies specifically designed around migraine neurobiology. It is increasingly used for chronic migraine, treatment-resistant migraine, and patients experiencing significant migraine-related disability.
Clinical experience suggests that some patients experience fewer migraine days, reduced acute medication use, and improved daily functioning during treatment. However, treatment response still varies considerably between individuals.
Erenumab: Clinical Data in Migraine
Several major clinical trials have evaluated erenumab efficacy in migraine prevention.
The STRIVE trial evaluated episodic migraine prevention in 955 patients and demonstrated significant reductions in monthly migraine days compared with placebo. The ARISE study also showed improvement in migraine frequency and functional outcomes among patients with episodic migraine.
The LIBERTY trial focused on patients who had failed multiple preventive therapies and demonstrated clinically meaningful benefit with erenumab treatment. Long-term extension studies have additionally suggested sustained migraine reduction and acceptable tolerability over multiple years.
These studies helped establish erenumab as one of the leading CGRP-targeting preventive therapies in migraine management.
Migraine-Related Disability and Quality of Life Scales in Erenumab Trials
Clinical trials evaluating erenumab frequently used disability and quality-of-life scales to assess broader patient outcomes. Common assessment tools included the Migraine Disability Assessment (MIDAS), Headache Impact Test (HIT-6), and Migraine-Specific Quality of Life Questionnaire (MSQ).
These measures help evaluate not only migraine frequency reduction but also productivity, emotional wellbeing, social participation, and day-to-day functioning.
CGRP Inhibitor Comparison Table
| Drug | Brand | Target | Dosing Frequency | Administration | FDA Approval | Key
Differentiator |
| Erenumab | Aimovig | CGRP receptor | Monthly | Subcutaneous injection | 2018 | Receptor-targeting mechanism |
| Fremanezumab | Ajovy | CGRP ligand | Monthly or quarterly | Subcutaneous injection | 2018 | Flexible dosing schedule |
| Galcanezumab | Emgality | CGRP ligand | Monthly | Subcutaneous injection | 2018 | Cluster headache indication |
| Eptinezumab | Vyepti | CGRP ligand | Quarterly | Intravenous infusion | 2020 | IV administration |
Common Side Effects of Erenumab
| Side Effect | Frequency | Management |
| Injection site reactions | Common | Rotate injection sites |
| Constipation | Common | Hydration and monitoring |
| Muscle cramps | Less common | Symptom management |
| Fatigue | Less common | Clinical evaluation |
| Hypertension | Reported in some patients | Blood pressure monitoring |
Erenumab has relatively limited known drug-drug interactions compared with many oral preventive migraine medications. However, clinicians still monitor concurrent neurological and cardiovascular therapies during treatment.
Safety and Tolerability of Erenumab
Most clinical trials suggest erenumab is generally well tolerated in many patients. Compared with older preventive therapies, CGRP inhibitors may produce fewer systemic neurological side effects such as sedation or cognitive slowing.
However, long-term safety monitoring remains important, particularly regarding cardiovascular risk, constipation severity, hypersensitivity reactions, and blood pressure changes.
Erenumab Storage Instructions
Erenumab autoinjectors should be refrigerated between 2°C and 8°C and protected from excessive heat, freezing, and direct light. Before injection, the medication may be left at room temperature for approximately 30 minutes.
Missed Dose & Contraindications
If a dose is missed, patients are generally advised to administer the injection as soon as possible and resume the monthly schedule from that date.
Erenumab should not be used in patients with known hypersensitivity to the medication or formulation components. Limited pregnancy safety data currently exists, so healthcare providers generally evaluate migraine severity and treatment necessity before use during pregnancy or lactation.
Erenumab Warning
Hypertension Warning : Post-marketing reports have identified elevated blood pressure in some patients receiving erenumab. Patients with uncontrolled hypertension or cardiovascular risk factors may require closer monitoring during treatment.
Read also about Hypertension in Pregnancy
Limitations in the Use of Erenumab
Despite strong clinical interest, erenumab has several limitations. Not all patients respond adequately to therapy, and some continue experiencing breakthrough migraine attacks despite treatment.
Additional challenges include high treatment cost, insurance authorization barriers, limited access in some countries, and uncertainty regarding very long-term biologic safety.
Aimovig (Erenumab) Availability by Country — 2026
In the United States, Aimovig received approval from the U.S. Food and Drug Administration (FDA) in 2018, with estimated monthly pricing ranging from approximately $575 to $700 before insurance coverage.
In the United Kingdom, the therapy is approved through NICE pathways for eligible migraine patients after failure of multiple preventive therapies.
Countries including France, Germany, and Italy also provide varying levels of reimbursement through national healthcare systems.
In India, erenumab availability remains more limited and is often accessed through import or specialty distribution pathways, with estimated monthly pricing ranging between ₹15,000 and ₹25,000.
List of Studies & Clinical Trials So Far
| Study | Population | Key Finding |
| STRIVE (2017) | Episodic migraine | Reduced monthly migraine days |
| ARISE (2018) | Episodic migraine | Improved migraine frequency |
| LIBERTY (2018) | Prior preventive failure | Significant clinical response |
| Open-label extension | Long-term follow-up | Sustained efficacy over years |
Erenumab: Future Directions
Future research surrounding erenumab is increasingly focused on long-term safety, comparative biologic effectiveness, biomarker-driven patient selection, and personalized migraine prevention strategies.
Researchers are also evaluating broader CGRP pathway biology and how migraine treatments may evolve within precision neurology frameworks.
Conclusions
Erenumab represents a major development in migraine prevention by targeting CGRP receptor pathways directly associated with migraine biology.
For many patients with chronic or treatment-resistant migraine, CGRP inhibitors such as Aimovig have expanded preventive treatment options beyond traditional therapies. However, treatment decisions still require individualized clinical evaluation based on migraine severity, treatment history, cardiovascular considerations, and patient-specific risk factors.
As migraine care becomes increasingly personalized, biologic therapies such as erenumab are likely to remain central to future preventive neurology strategies.
Explore healthcare insights and migraine-related healthcare perspectives at MDForLives . Join global healthcare conversations and healthcare research communities
Also Join Migraine Patient Community
Frequently Asked Questions
What is the success rate of erenumab?
Clinical trials demonstrated meaningful migraine reduction in many patients, although response rates vary depending on migraine severity and prior treatment history.
How long does it take for erenumab to work?
Some patients notice improvement within the first month, while others may require several months for full therapeutic effect.
How long does erenumab last?
Erenumab is designed for monthly administration due to its long biologic half-life.
Does erenumab cause hair loss?
Hair loss has been reported anecdotally in some patients, although it is not considered one of the most common adverse effects identified in major trials.
What is the best medication for chronic migraines?
The best medication depends on migraine frequency, comorbidities, prior treatment response, and patient-specific clinical factors. CGRP inhibitors, including erenumab, are increasingly used for preventive treatment.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.