Eye gene therapy is emerging as a targeted approach to treat inherited retinal diseases by correcting the genetic defects that cause vision loss. Unlike conventional treatments that manage symptoms, this method works at a molecular level to restore or preserve visual function. As clinical applications expand, understanding how it works and who it benefits is becoming increasingly relevant.
What is Eye Gene Therapy?
Overview
Eye gene therapy involves delivering functional genetic material into retinal cells to compensate for defective or missing genes. The retina is a suitable target because of its accessibility and well-defined structure.
Types of Eye Gene Therapy
Different approaches are used depending on the disease:
- Gene replacement to restore missing gene function
- Gene silencing to block harmful gene activity
- Gene editing to correct mutations directly
Each approach is selected based on the underlying genetic mechanism.
History of Gene Therapy Development for Retinal Diseases
Early research in retinal gene therapy focused on rare inherited disorders with limited treatment options. Advances in viral vector design improved delivery efficiency, enabling more consistent outcomes.
Clinical breakthroughs over the past decade have shifted the field from experimental trials to approved therapies, particularly in inherited retinal conditions.
Read also about Gene Therapy
What Eye Diseases Can Be Treated with Gene Therapy?
Inherited Retinal Diseases
These include conditions such as retinitis pigmentosa and Leber congenital amaurosis, where specific gene mutations lead to progressive vision loss.
Role of RPE65 Mutation
One of the most studied mutations involves the RPE65 gene. Defects in this gene impair the visual cycle, leading to early-onset blindness. Targeting this mutation has led to successful therapeutic development.
Gene Therapy Approaches for Retinal Diseases
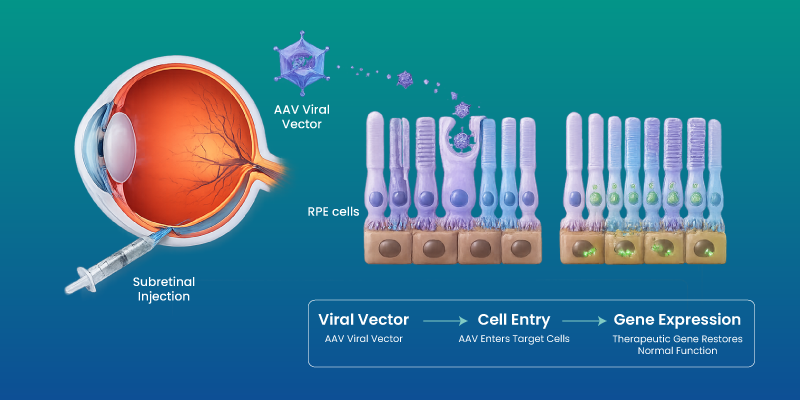
Delivery typically involves viral vectors, most commonly adeno-associated viruses, which transport the therapeutic gene into retinal cells.
The process ensures that corrected genetic instructions are expressed within targeted cells, enabling restoration of function where possible.
Luxturna: The First FDA-Approved Eye Gene Therapy
Luxturna represents a milestone in retinal gene therapy. It targets RPE65 mutations and has demonstrated improvement in functional vision in treated patients.
This approval established proof of concept for gene-based treatments in ophthalmology and paved the way for further development.
Ongoing Retinal Gene Therapy Clinical Development
Current research is expanding into broader indications.
Clinical trials are investigating:
- Additional inherited retinal diseases
- Improved delivery systems
- Longer-lasting therapeutic effects
These studies aim to extend benefits beyond a limited number of genetic conditions.
Approved Gene Therapy for Retinal Diseases
While Luxturna remains a key example, additional therapies are under evaluation. Regulatory approvals are expected to increase as evidence accumulates.
This gradual expansion reflects both scientific progress and regulatory caution.
Benefits and Effectiveness of Eye Gene Therapy
Gene therapy offers distinct clinical advantages.
| Benefit | Outcome |
| Targeted treatment | Addresses genetic cause directly |
| Potential long-term effect | Reduces need for repeated interventions |
| Improved function | Enhances visual capability in some patients |
Effectiveness depends on disease stage and timing of intervention.

Risks and Side Effects
Common Side Effects
Most side effects are procedure-related and may include:
- Mild inflammation
- Temporary vision disturbance
- Eye discomfort
Serious Risks
Although less common, potential risks include:
- Retinal damage
- Infection
- Immune response to vector
Careful patient selection and monitoring reduce these risks.
Retinal Gene Therapy Challenges
Several challenges limit widespread adoption.
- Genetic variability across patients
- Difficulty targeting advanced disease stages
- High cost of development and treatment
These factors influence both accessibility and scalability.
Cost and Accessibility of Eye Gene Therapy
Gene therapy treatments are expensive due to complex manufacturing and delivery processes.
Access varies across regions, with availability often limited to specialized centers. Insurance coverage and reimbursement models are still evolving.
Eye Gene Therapy Procedure, Recovery & Patient Journey
The procedure involves delivering the therapeutic gene directly into the retina through a surgical approach.
Recovery typically includes:
- Short-term monitoring for complications
- Gradual improvement in visual function
- Follow-up assessments to evaluate outcomes
The patient journey requires coordination between ophthalmologists and genetic specialists.
Who Should Consider Eye Gene Therapy?
Candidates are typically individuals with confirmed genetic mutations linked to retinal disease.
Eligibility depends on:
- Type of mutation
- Stage of disease
- Overall eye health
Early diagnosis improves the likelihood of positive outcomes.
Future of Eye Gene Therapy
Future developments are focused on expanding treatment scope and improving delivery methods.

Key directions include:
- Gene editing technologies
- Broader application across retinal conditions
- Improved vector design for better targeting
These advances aim to make therapy more accessible and effective.
Read also about CRISPR Gene Editing
Conclusion
Eye gene therapy represents a shift from symptom management to addressing the genetic basis of vision loss. While current applications are limited to specific conditions, ongoing research is expanding its potential. As technology advances, it is likely to become a key component of ophthalmic care.
Explore more evidence-led healthcare insights: MDForLives
Frequently Asked Questions
What is the success rate of eye gene therapy?
Success varies by condition, but clinical studies show meaningful improvement in selected patients.
Can gene therapy make vision worse?
There is a risk of complications, but careful monitoring reduces the likelihood of adverse outcomes.
Is eye gene therapy a permanent cure?
It may provide long-term benefit, but durability depends on disease and treatment response.
How long is recovery after eye gene therapy?
Recovery varies, but initial healing occurs within weeks, followed by longer-term functional improvement.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.





2 Comments
Charcot Marie Tooth disease therapy: Phase III trial has begun
5 years ago[…] motor sensory neuropathy (HMSN). Patients can carry duplications, mutations, or other changes in genes that lead to abnormalities in nerve axons, problems with myelination, or both. Peripheral nerves […]
Phase III trial beginning for a Charcot-Marie-Tooth disease therapy - MDForLives
5 years ago[…] motor sensory neuropathy (HMSN). Patients can carry duplications, mutations, or other changes in genes that lead to abnormalities in nerve axons, problems with myelination, or both. Peripheral nerves […]