

GLP-1 Therapy in Obesity Care: Why Real-World Continuation Still Falls Short
Most clinicians treating obesity today are not debating whether GLP-1 therapies work. That question has largely been answered. Across markets, clinical trials, guidelines, and day-to-day experience have established their role in weight management.
The more uncomfortable question is whether starting these therapies always makes sense when continuation cannot be guaranteed.
This tension shows up quietly in practice. A therapy can be clinically appropriate, evidence-backed, and aligned with guidelines, yet still feel difficult to initiate. Not because of doubt in the molecule, but because of uncertainty around everything that comes after.
That is where GLP-1 therapy sits in obesity care today: at the intersection of high clinical confidence and fragile real-world continuity.
These observations reflect how GLP‑1 therapy is being evaluated and experienced in everyday clinical practice.
NOTE: This article is based on clinician-reported research conducted across the USA, UK, Canada, France, Germany, and Italy.
Has GLP-1 Therapy Become Clinically Established in Obesity Care?
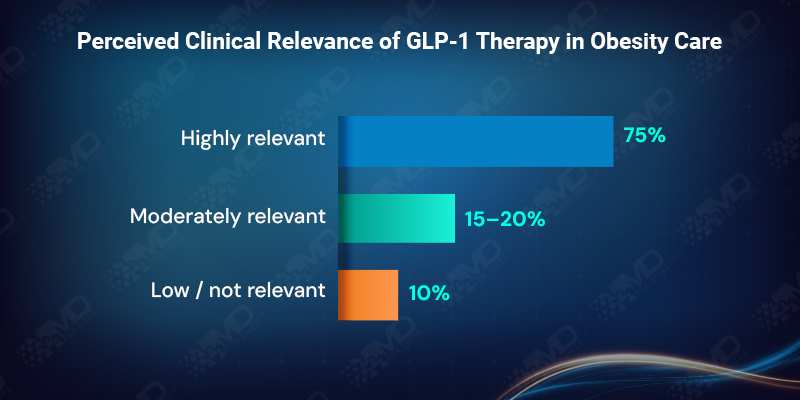
GLP-1 therapy has crossed a meaningful threshold in how clinicians view obesity management. More than 90% of clinicians describe GLP-1 or GLP-1/GIP therapies as either highly or moderately relevant to their role in treating obesity, with nearly three-quarters rating them as highly relevant.
This level of agreement signals that GLP-1 therapy is no longer seen as optional, experimental, or peripheral. It has become part of the standard mental framework for obesity care. The remaining hesitation does not stem from uncertainty about efficacy. It stems from whether long-term use can realistically be supported once treatment begins.
In other words, belief has stabilized. Practical confidence has not.
How System Readiness Shapes Routine Consideration of GLP-1 Therapy?
At an overall level, around two-thirds of clinicians say they routinely consider GLP-1 therapy for obesity independent of diabetes, while another quarter consider it occasionally. On the surface, this suggests widespread adoption.
But market-level patterns reveal a more nuanced reality.
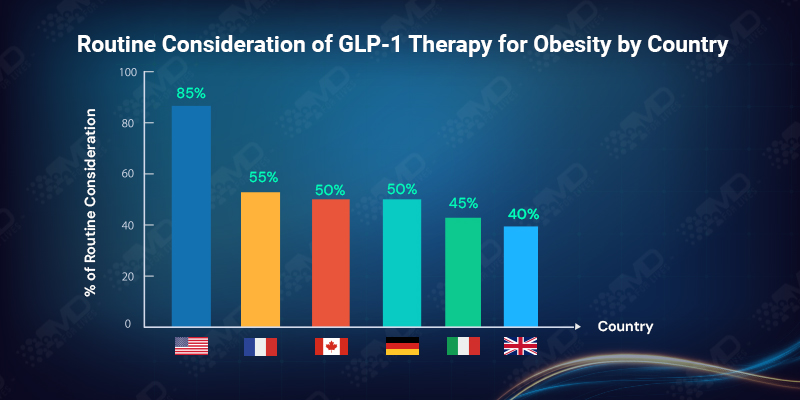
In the USA, more than 80% of clinicians report routine consideration, reflecting fewer formal barriers at the point of prescribing. In contrast, in the UK, fewer than 40% describe GLP-1 therapy as a routine option, with the majority indicating only occasional consideration.
This divergence is not driven by differences in belief. It reflects how access pathways, referral requirements, and prescribing controls shape behavior. Where systems are permissive, GLP-1 becomes routine. Where systems are restrictive, clinicians adapt by reserving therapy for specific situations, even when they believe it could be beneficial.
Relevance, in practice, is filtered through system design.
Why Real-World GLP-1 Outcomes Erode Despite Strong Trial Alignment?
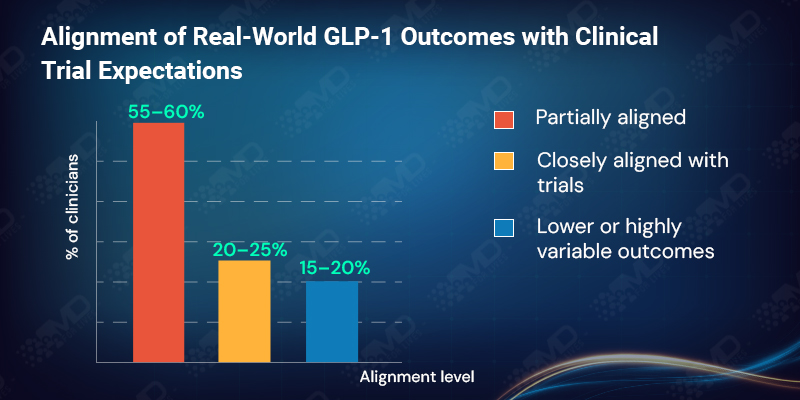
Confidence in GLP-1 outcomes remains high, but it is measured rather than absolute. More than 80% of clinicians report that real-world weight loss outcomes are aligned with clinical trial expectations, yet the majority describe this alignment as only partial rather than close.
Around 15% indicate that outcomes are often lower than expected or highly variable. Importantly, clinicians do not interpret this as drug failure. They interpret it as the consequence of real-world interference: interrupted treatment, delayed titration, coverage gaps, and early discontinuation.
The result is a subtle shift in mindset. GLP-1 efficacy is trusted, but durability is no longer assumed. Continuity, rather than pharmacology, is seen as the deciding factor in whether outcomes hold.
How Clinicians Are Selecting Patients When Long-Term Feasibility Is Uncertain?
When clinicians describe how they assess suitability for GLP-1 therapy, clinical eligibility and comorbidities remain the primary driver, cited by just over half of respondents. However, nearly one-third now say expected adherence and patient motivation weigh most heavily in their decisions.
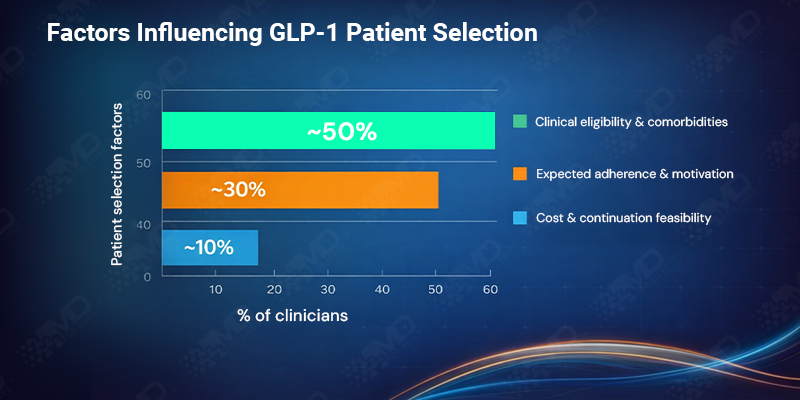
Cost and coverage are less frequently named as the single dominant factor, influencing around 10% directly. Yet their indirect influence is clear. Clinicians are increasingly selecting patients based not only on who qualifies at baseline, but on who can realistically remain on therapy.
This introduces a quiet shift in obesity care. Some patients may never start GLP-1 therapy, not because they are clinically unsuitable, but because continuation feels uncertain from the outset.
What Clinicians Cite as the Leading Drivers of GLP-1 Discontinuation?
When GLP-1 therapy is stopped, the reason is rarely unexpected. Across markets, more than half of clinicians identify cost or loss of coverage as the primary limiter of long-term use. Gastrointestinal side effects account for just under 30%, while adherence issues and clinical reassessment together make up a much smaller share.
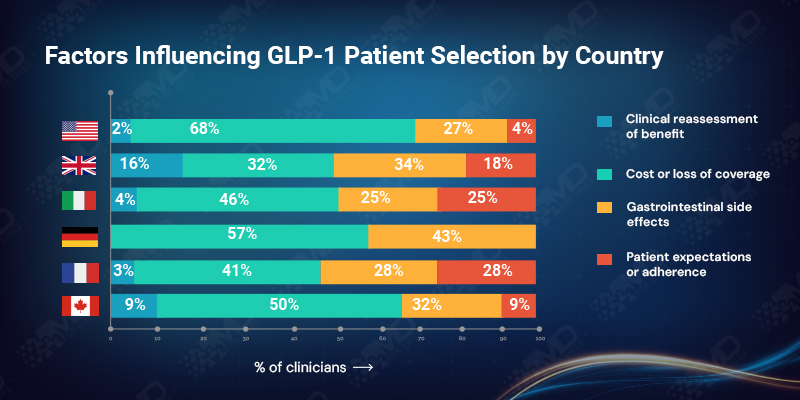
The pattern is especially stark in the USA, where nearly 70% cite financial disruption as the leading cause of discontinuation. In the UK, the picture is more distributed, with side effects, system review, and reassessment playing a larger role alongside access constraints.
What unites these experiences is that discontinuation is rarely driven by lack of clinical benefit. Clinicians are generally comfortable managing tolerability. What they struggle with is stopping a therapy that is working for reasons unrelated to response.
That experience feeds back into future prescribing decisions, making clinicians more cautious about initiating what they may not be able to sustain.

“The clinical decision is usually clear. Managing everything around it is where the work really begins.”
Dr. Michael R. Lawson, MD, Endocrinologist ,United States
Why Chronic Obesity Management Remains Uneven Across Care Settings?
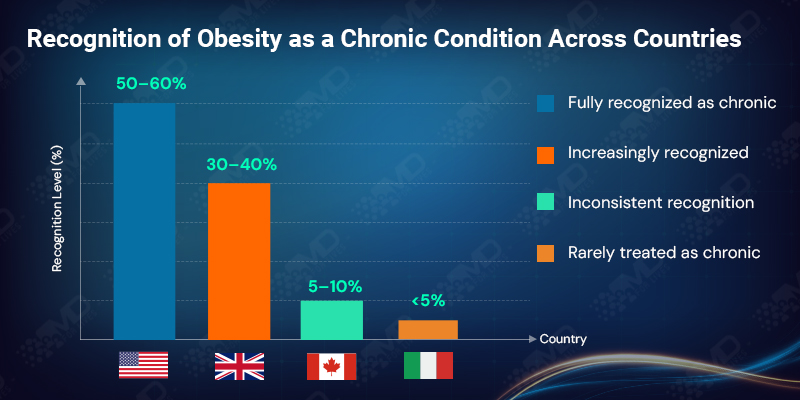
Most clinicians report that obesity is either fully recognized as a chronic condition (just over 55%) or increasingly recognized as such (around 35%). This shift aligns with the rise of long-term pharmacologic approaches, including GLP-1 therapy.
Yet close to 10% still describe obesity care as inconsistent, and a small fraction say it is rarely treated as chronic at all. This inconsistency matters. GLP-1 therapy requires sustained engagement, long-term expectation setting, and continuity of care. Where obesity is still approached episodically, treatment interruptions become more likely.
In several markets, therapeutic innovation appears to be moving faster than cultural change in care models.
How Access Barriers Differ by Market but Produce Similar Outcomes?
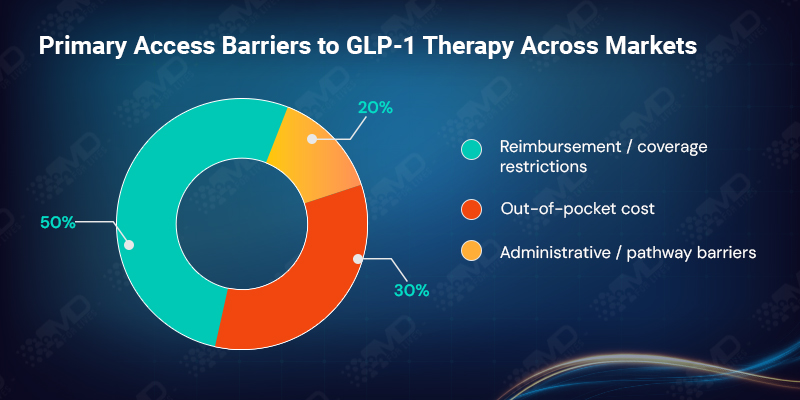
Across regions, access constraints fall into two dominant categories. Nearly half of clinicians cite reimbursement or coverage restrictions as the primary barrier to GLP-1 access, while just over 30% point to out-of-pocket cost. Administrative hurdles and limited care pathways account for the remainder.
The balance between these barriers varies by system. In the USA, patient cost exposure dominates. In the UK, prescribing restrictions and pathway limitations are more visible. In Italy and Germany, regional or statutory reimbursement decisions play a decisive role. In Canada, provincial variation and authorization processes fragment access.
Despite these differences, the outcome is consistent. Access remains unpredictable, and continuity is difficult to guarantee. Clinicians adjust conversations and prescribing behavior, accordingly, often factoring anticipated payer response into clinical discussions.
Why Patient Expectations Remain Misaligned with Long-Term GLP-1 Use?
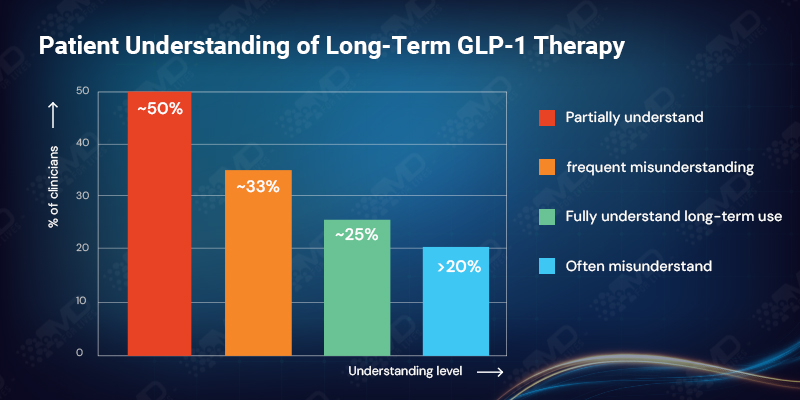
Only about one-quarter of clinicians believe patients generally understand the long-term nature of GLP-1 therapy. The majority describe understanding as partial, while more than 20% say expectations are often misunderstood.
This misalignment adds friction at multiple points. Patients may anticipate short-term treatment or rapid results, while clinicians are planning for sustained use. When access disruptions or side effects emerge, motivation drops more quickly if expectations were never fully aligned.
In the UK, expectation misalignment is particularly visible, with roughly one-third of clinicians reporting frequent misunderstanding. Across markets, expectation setting emerges as one of the most fragile points in the GLP-1 treatment journey.

“Patients often come in thinking short term. As clinicians, we’re already planning for long-term care.”
Dr. James Whitmore, MD, Senior Endocrinologist, United Kingdom
What Will Ultimately Determine the Real-World Impact of GLP-1 Therapy?
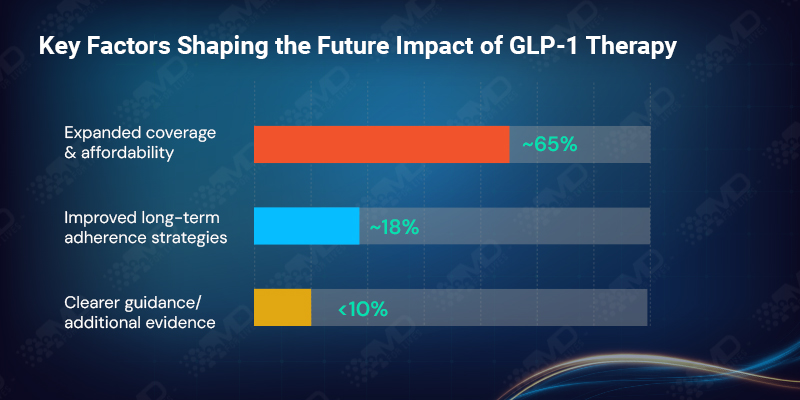
When clinicians look ahead, their priorities are clear. Nearly 65% identify expanded coverage and affordability as the single most important factor shaping the future impact of GLP-1 therapy in obesity care. Another 18% emphasize the need for better long-term adherence strategies.
Clearer guidance and additional real-world evidence rank far lower. This is telling. For most clinicians, the science has already done its job. What remains unresolved is whether healthcare systems can support sustained use at scale.
In the USA, affordability dominates future expectations. In the UK, clearer guidance carries more weight, reflecting the influence of centralized decision-making and prescribing frameworks.
Closing Perspective
GLP-1 therapy represents one of the most significant advances in obesity treatment in recent years. Among clinicians, its role is no longer in question. Initiation is broadly accepted; efficacy is trusted, and obesity is increasingly framed as a chronic condition.
What limits real-world impact is not uncertainty about the therapy itself, but the fragility of the system that surrounds it. Coverage volatility, cost exposure, uneven pathways, and misaligned expectations consistently undermine long-term use.
GLP-1 therapy has crossed the belief threshold. It has not yet crossed the delivery threshold. Until access frameworks, reimbursement models, and patient expectations align with chronic disease management, GLP-1 will continue to sit at the intersection of clinical confidence and system friction.
MDForLives is a global healthcare focused insights platform where healthcare professionals, patients, and caregivers discover, share, and shape the future of healthcare.
We turn real opinions and verified data into authentic healthcare insights and intelligence.
Hope this insight added value.
Participate in the survey below and add your voice to the conversation.
About Author : MDForlives
MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.



