Laser eye surgery risks are an important consideration for anyone thinking about vision correction procedures such as LASIK. While LASIK eye surgery has helped millions of people reduce their dependence on glasses and contact lenses, the procedure can involve potential side effects and complications. Understanding LASIK eye surgery side effects, complications, and long term risks helps patients make informed decisions about whether laser vision correction is appropriate for their needs.
What Is LASIK Eye Surgery?
LASIK (Laser Assisted In Situ Keratomileusis) is a surgical procedure used to correct refractive vision problems such as:
- Nearsightedness (myopia)
- Farsightedness (hyperopia)
- Astigmatism
The procedure reshapes the cornea using a specialized laser so that light entering the eye focuses properly on the retina.
How It Works
During LASIK surgery, an ophthalmologist creates a thin flap in the cornea. A laser then reshapes the underlying corneal tissue to correct vision.
The flap is repositioned afterward and typically heals without stitches.
Most procedures take less than 30 minutes and are performed on an outpatient basis.
Why People Choose LASIK
Many people choose LASIK because it offers several potential benefits:
- Reduced dependence on glasses or contact lenses
- Quick recovery time
- Long lasting vision correction
- High patient satisfaction rates
However, it is still important to understand the laser eye surgery side effects and risks before undergoing the procedure.
Is LASIK Safe?
LASIK surgery is widely performed and has been studied extensively.
Approval and Safety Standards
In the United States, LASIK technology and devices are regulated by the U.S. Food and Drug Administration. Medical device approvals require extensive testing to demonstrate safety and effectiveness.
Medical Consensus
Organizations such as the American Academy of Ophthalmology state that LASIK can be safe and effective for appropriately selected patients.
However, like any surgical procedure, LASIK carries certain risks and potential complications.
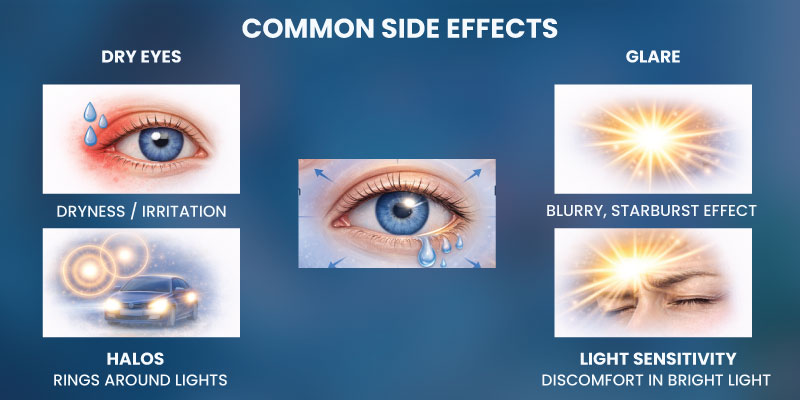
Common Short Term Side Effects of LASIK
Most LASIK patients experience mild temporary symptoms during the healing process.
Common laser eye surgery side effects include:
- Dry eyes
- Light sensitivity
- Blurry vision during early recovery
- Eye irritation or discomfort
- Glare or halos around lights
These symptoms often improve within weeks or months after surgery.
Serious LASIK Complications
Although uncommon, some patients may experience more significant complications.
Possible LASIK eye surgery complications include:
- Corneal flap complications
- Infection or inflammation
- Overcorrection or undercorrection of vision
- Corneal ectasia (corneal weakening)
- Persistent dry eye syndrome
Serious complications are relatively rare but may require additional treatment or corrective procedures.
Long Term Risks of LASIK Eye Surgery
Some LASIK side effects may persist long term in certain patients.
Potential long term LASIK risks include:
- Chronic dry eyes
- Vision fluctuations
- Difficulty with night driving
- Reduced contrast sensitivity
Long term outcomes often depend on individual factors such as eye health and surgical technique.
Night Vision Problems After LASIK
Some patients report night vision problems after LASIK, particularly during the first few months following surgery.
Symptoms may include:
- Halos around lights
- Starbursts
- Increased glare while driving at night
In many cases these issues improve as the eye heals.

Who Is at Higher Risk of LASIK Complications?
Not everyone is an ideal candidate for LASIK surgery.
Higher risk groups may include individuals with:
- Thin corneas
- Severe dry eye syndrome
- Unstable vision prescriptions
- Certain autoimmune conditions
- Corneal diseases
A comprehensive eye examination helps determine whether LASIK is appropriate.
LASIK Eye Surgery Risks vs Benefits
Understanding the balance between risks and benefits is essential.
Benefits may include:
- Improved visual clarity
- Reduced reliance on corrective lenses
- Faster visual recovery compared with some procedures
Potential risks include:
- Surgical complications
- Long term visual disturbances
- Need for additional corrective procedures
Careful evaluation by an ophthalmologist helps determine the best treatment option.
Real Patient Experiences and LASIK Regret
Most LASIK patients report satisfaction with their results. However, some individuals may experience unexpected outcomes.
LASIK regret may occur when:
- Visual expectations are not met
- Side effects persist longer than expected
- Additional procedures are required
Clear communication with surgeons before surgery helps manage expectations.
How to Reduce LASIK Eye Surgery Risks
Patients can reduce laser eye surgery risks by following several important steps.
Recommended precautions include:
- Choosing an experienced ophthalmologist
- Undergoing comprehensive pre surgery testing
- Discussing medical history with the surgeon
- Following post surgery care instructions carefully
Proper screening significantly lowers the risk of complications.
Alternatives to LASIK Eye Surgery
Individuals who are not suitable candidates for LASIK may consider alternative procedures.
Possible alternatives include:
- PRK (Photorefractive Keratectomy)
- SMILE eye surgery
- Implantable contact lenses (ICL)
- Continued use of glasses or contact lenses
An eye specialist can recommend the most appropriate option.
Cost vs Risk Considerations
LASIK surgery costs vary depending on location, technology, and surgeon experience.
While cost may influence decision making, safety and medical suitability should remain the primary considerations.
Patients should prioritize qualified surgeons and reputable clinics over lower priced procedures.
Conclusion
LASIK eye surgery has helped millions of people improve vision and reduce dependence on corrective lenses. However, understanding laser eye surgery risks, side effects, and complications is essential before choosing the procedure.
By consulting experienced ophthalmologists, evaluating candidacy carefully, and considering alternative treatments when necessary, patients can make informed decisions about their eye health.
Read also about Cataract Surgery
Frequently Asked Questions
Are there any permanent side effects of LASIK?
Some patients may experience long term effects such as dry eyes or mild visual disturbances. However, most LASIK side effects are temporary and improve over time.
Is LASIK safe in the long term?
For most properly selected patients, LASIK is considered safe with stable long term outcomes. Regular eye checkups remain important after surgery.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.