Colorectal cancer research is rapidly transforming how scientists understand, detect, and treat one of the most common cancers worldwide. Advances in medical science, including precision medicine, immunotherapy, and artificial intelligence driven diagnostics, are improving early detection and treatment outcomes. Ongoing colorectal cancer clinical research continues to explore innovative therapies and prevention strategies aimed at increasing survival rates and improving quality of life for patients.
Why Colorectal Cancer Research Matters?
Research plays a critical role in improving outcomes for patients diagnosed with colorectal cancer. Through ongoing studies and clinical trials, scientists are discovering new ways to detect the disease earlier, develop more effective treatments, and reduce treatment related side effects.
Improving Survival Rates
Medical research has significantly improved survival rates for colorectal cancer over the past decades. Early detection combined with advanced treatment approaches has helped many patients achieve better outcomes.
Scientists continue to study how genetic mutations, lifestyle factors, and tumor biology influence disease progression and response to treatment.
Early Detection and Screening Advances
Screening programs have become one of the most effective strategies in preventing colorectal cancer deaths. Modern screening technologies include:
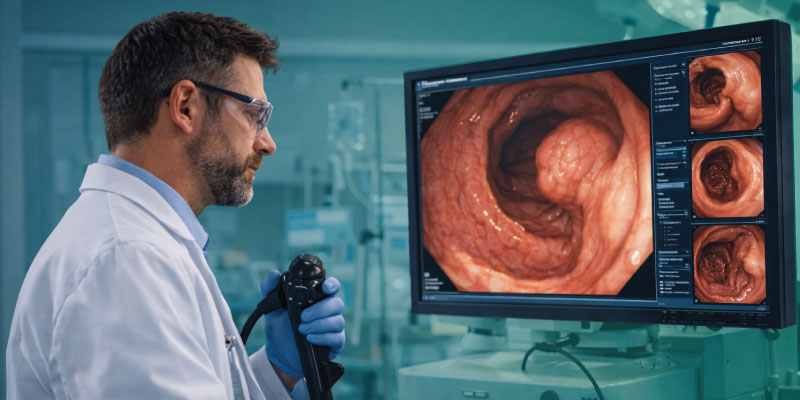
- Colonoscopy screening
- Stool based DNA tests
- Advanced imaging techniques
Research is also exploring AI driven diagnostic tools that can identify abnormal tissue during screening procedures with higher accuracy.
Reducing Treatment Side Effects
Traditional cancer therapies often cause significant side effects. Research now focuses on developing targeted therapies that attack cancer cells while minimizing damage to healthy tissue.
These innovations aim to improve patient quality of life during treatment.
Latest Breakthroughs in Colorectal Cancer Research
Scientific progress has introduced several promising developments in colorectal cancer treatment and diagnosis.
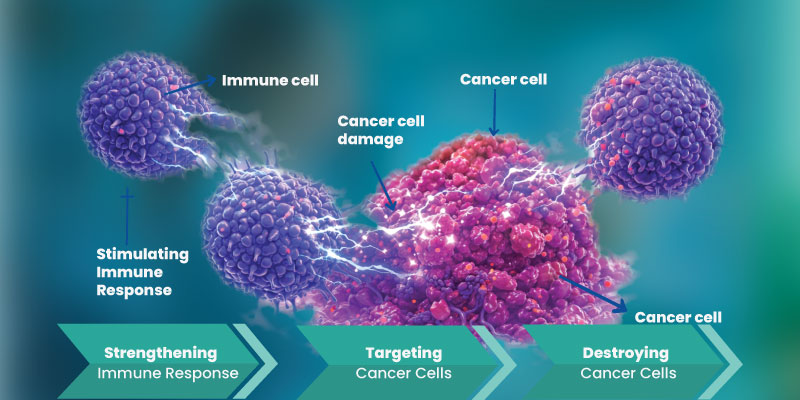
Immunotherapy for Colorectal Cancer
Immunotherapy treatments stimulate the immune system to recognize and destroy cancer cells. Certain colorectal cancers respond well to immune checkpoint inhibitors that help the immune system attack tumors more effectively.
Researchers continue to study which patients benefit most from immunotherapy treatments.
Targeted Therapy and Precision Medicine
Targeted therapies focus on specific genetic mutations present in cancer cells. By identifying these molecular changes, doctors can select treatments designed to attack those specific cancer pathways.
Precision medicine approaches allow doctors to tailor treatment strategies for individual patients.
Advances in Genetic Testing
Genetic testing plays an increasingly important role in colorectal cancer care. Researchers are studying genetic markers that may help predict cancer risk, treatment response, and recurrence probability.
These discoveries may help physicians personalize treatment plans and identify high risk individuals earlier.
AI and Early Detection Research
Artificial intelligence is being integrated into medical imaging and diagnostic systems. AI algorithms can analyze colonoscopy images and pathology data to detect abnormal tissue patterns.
These technologies have the potential to improve early detection rates and reduce missed diagnoses.
Clinical Trials in Colorectal Cancer Research
Clinical trials play a vital role in developing new treatments for colorectal cancer. Through carefully controlled studies, researchers test the safety and effectiveness of innovative therapies.
Patients participating in clinical trials may gain access to new treatments before they become widely available.
Clinical research continues to explore:
- Novel immunotherapy combinations
- New chemotherapy regimens
- Targeted drug therapies
- Personalized treatment approaches
These studies help determine which treatments offer the greatest benefit to patients.
New Treatment Options Emerging from Research
Ongoing scientific discoveries continue to shape new treatment approaches for colorectal cancer.
Minimally Invasive Surgery
Advances in surgical techniques allow doctors to remove tumors with smaller incisions and shorter recovery times.
Minimally invasive procedures can reduce complications and improve patient recovery.
Combination Therapies
Researchers are studying combinations of treatments such as chemotherapy, targeted therapy, and immunotherapy.
Combining therapies may enhance treatment effectiveness and reduce the likelihood of cancer resistance.
Personalized Treatment Plans
Precision medicine allows oncologists to design treatment strategies based on a patient’s unique tumor genetics and medical history.
Personalized care helps maximize treatment success while minimizing unnecessary therapies.
What Are the Advancements in Colorectal Cancer Treatment?
Recent advancements include:
- Immunotherapy drugs targeting specific tumor pathways
- Improved surgical techniques
- AI assisted diagnostics
- Genetic profiling for treatment planning
These innovations represent a major shift toward individualized cancer care.
Colorectal Cancer Prevention Research and Lifestyle Studies
Preventive research continues to explore how lifestyle and environmental factors influence colorectal cancer risk.
Diet and Colorectal Cancer Risk
Dietary patterns play an important role in colorectal cancer prevention. Research suggests that diets rich in fiber, fruits, vegetables, and whole grains may reduce cancer risk.
Conversely, high consumption of processed meats and low fiber diets may increase risk.
Exercise and Cancer Prevention
Regular physical activity has been linked to lower colorectal cancer risk. Exercise supports healthy metabolism and reduces inflammation associated with cancer development.
Aspirin and Chemoprevention Studies
Researchers are studying whether long term use of low dose aspirin may reduce colorectal cancer risk in certain populations. These studies aim to determine the balance between benefits and potential risks.
How Does Colorectal Cancer Research Affect Long-Term Survival and Life Expectancy?
Research advancements contribute directly to improved survival rates and better long term outcomes for patients.
Improved screening programs allow earlier diagnosis, while modern treatments provide more effective tumor control. Personalized medicine and targeted therapies continue to extend survival and improve patient quality of life.
As research progresses, the outlook for colorectal cancer patients continues to improve.
Conclusion
Colorectal cancer research continues to drive major advances in prevention, diagnosis, and treatment. From immunotherapy and precision medicine to AI assisted screening technologies, modern research is reshaping the future of colorectal cancer care.
Continued investment in research and clinical trials will remain essential for improving survival rates and reducing the global burden of colorectal cancer.
Read also about Cervical Cancer Prevention
Frequently Asked Questions
What are the early signs and symptoms that should be watched for?
Early signs of colorectal cancer may include changes in bowel habits, blood in the stool, abdominal discomfort, unexplained weight loss, and persistent fatigue.
Are there new colorectal cancer treatments that improve survival or reduce side effects?
Yes. Advances in immunotherapy, targeted therapy, and minimally invasive surgery are improving treatment outcomes while reducing treatment related side effects.
What can be done to lower risk or prevent recurrence?
Preventive measures may include regular screening, maintaining a healthy diet, exercising regularly, avoiding smoking, and following medical guidance for cancer prevention.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.