Two patients can receive the same diagnosis and respond very differently to treatment. One improves quickly. The other experiences limited benefit or significant side effects. Precision medicine addresses this gap by tailoring treatment decisions to biological differences rather than relying only on population averages.
By combining genomic testing, biomarkers, and clinical data, precision medicine helps clinicians select therapies that are more likely to work for specific patient groups. It is increasingly used in oncology, cardiology, rare diseases, and pharmacology.
Key Takeaways
- Precision medicine uses genetic and molecular data to guide treatment decisions.
- It improves therapy selection in diseases with identifiable biomarkers.
- It reduces unnecessary exposure to ineffective treatments.
- Access, cost, and data interpretation remain important challenges.
Precision Medicine Overview
Precision medicine is a medical approach that uses genetic, molecular, environmental, and lifestyle information to guide prevention and treatment strategies for defined patient groups.
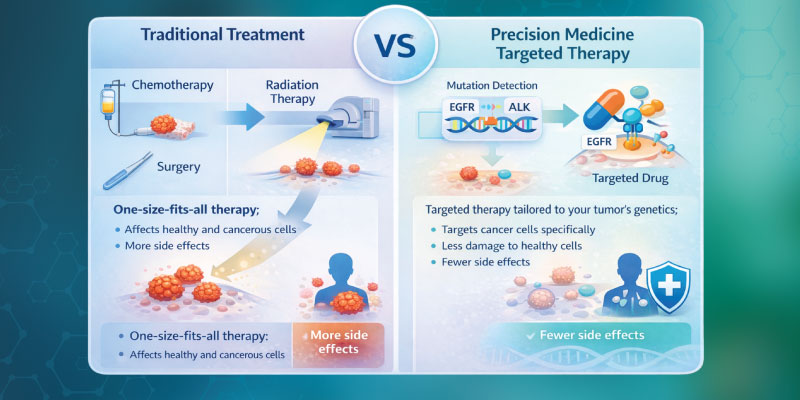
Traditional treatment models rely on broad clinical guidelines developed from large populations. Precision medicine identifies subgroups within those populations who are more likely to respond to specific therapies.
The term is often used alongside personalized medicine. Precision medicine focuses on stratifying patients into biologically defined groups. Personalized medicine implies fully individualized care, which is not always feasible in clinical practice.
How Does Precision Medicine Work?
Precision medicine integrates several data sources:
- Genomic sequencing to identify mutations or inherited variants
- Biomarker testing to detect molecular signals linked to treatment response
- Pharmacogenomic profiling to predict drug metabolism
- Clinical data analysis including imaging and laboratory results
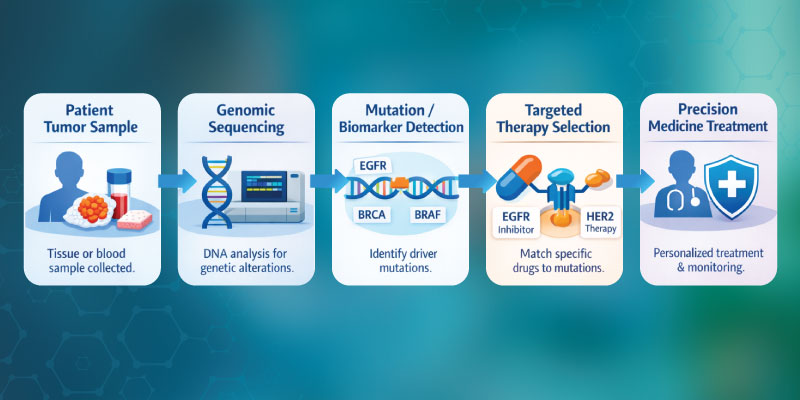
For example, in lung cancer, testing may reveal an EGFR mutation. If present, a targeted therapy designed for that mutation can be prescribed. Without the mutation, the same drug would likely provide little benefit.
This approach reduces trial and error prescribing and improves treatment alignment.
Read also about medication errors
Clinical Applications of Precision Medicine
Oncology
Cancer care represents the most established use of precision medicine. HER2 targeted therapy in breast cancer and EGFR inhibitors in lung cancer are widely adopted examples.
In cancers with actionable mutations, targeted therapies have been associated with improved progression free survival in selected patient groups compared to non targeted approaches.
Pharmacogenomics
Genetic variations in enzymes such as CYP2C19 and CYP2D6 influence how patients metabolize medications. Testing can guide safer dosing of antidepressants, anticoagulants, and cardiovascular drugs.
Rare Genetic Disorders
Whole exome sequencing can identify causative variants, allowing earlier intervention and targeted monitoring.
Cardiovascular Risk Stratification
Biomarker based assessment helps identify patients at elevated risk and supports preventive strategies.
Key Components and Tools of Precision Medicine
| Tool | What It Identifies | Clinical Use |
| Genomic sequencing | DNA mutations or variants | Targeted therapy selection |
| Biomarker assays | Molecular or protein markers | Predict treatment response |
| Companion diagnostics | Drug eligibility criteria | Regulatory aligned prescribing |
| Pharmacogenomic testing | Drug metabolism patterns | Safer dosing |
| Clinical decision systems | Integrated risk patterns | Treatment planning support |
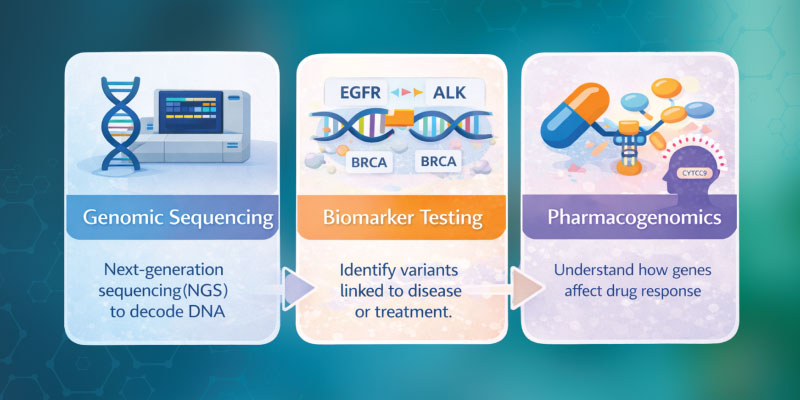
These tools allow clinicians to make evidence informed decisions based on measurable biological differences.
Benefits of Precision Medicine for Patients
Precision medicine offers several advantages when applicable:
- More accurate therapy matching
- Reduced adverse drug reactions
- Earlier identification of disease risk
- Better stratification in clinical trials
- Improved understanding of disease subtypes
It is most effective in conditions with clearly defined molecular drivers.
Challenges and Limitations of Precision Medicine
Despite progress, limitations remain:
- Not all diseases have actionable biomarkers
- Some genetic findings are classified as variants of uncertain significance
- Access to genomic testing varies
- Costs may be high
- Data privacy and governance require careful oversight
Precision medicine enhances care in specific contexts. It does not replace all traditional treatment models.
Future Directions in Precision Medicine
Emerging developments include:
- Integration of multi omics data
- Artificial intelligence assisted genomic interpretation
- Expansion beyond oncology into metabolic and autoimmune diseases
- Broader representation in genomic databases
As data quality improves and testing becomes more accessible, precision approaches are expected to expand further into routine care.
Conclusion
Precision medicine represents an evolution in how treatment decisions are made. By aligning therapy with genetic and molecular characteristics, it improves targeting accuracy and supports patient centered care.
Its success depends on evidence, appropriate patient selection, equitable access, and responsible data management. When applied correctly, precision medicine strengthens the connection between biological insight and clinical outcomes.
Read also about
Frequently Asked Questions
What is the main difference between precision medicine and regular treatment?
Traditional treatment applies standardized therapies based on population averages. Precision medicine uses genetic and molecular information to guide therapy selection for defined patient groups.
Does precision medicine work for all cancers?
No. Precision medicine is most effective when cancers have identifiable and actionable mutations. Many tumors still lack validated molecular targets.
What are examples of precision medicine?
Examples include HER2 targeted therapy in breast cancer, EGFR inhibitors in lung cancer, pharmacogenomic guided antidepressant dosing, and CAR T cell therapy.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.