Biosimilars are reshaping treatment access by offering clinically comparable alternatives to biologic therapies. As healthcare systems manage rising costs and demand for advanced treatments, they are becoming central to improving affordability without compromising clinical outcomes.
What Are Biosimilars?
Biosimilar Drugs
Biosimilars drugs are biologic products designed to be highly similar to an already approved reference biologic. They match in safety, efficacy, and quality within defined regulatory standards.
What Are Biologic Medicines?
Biologic medicines are complex therapies derived from living cells. They are used to treat conditions such as cancer, autoimmune disorders, and chronic inflammatory diseases.
Read also about Precision Medicine
Biologics and Biosimilars Explained
While biologics are original products, they are developed after patent expiry. Due to their biological origin, exact replication is not possible, but clinical equivalence is established through rigorous testing.
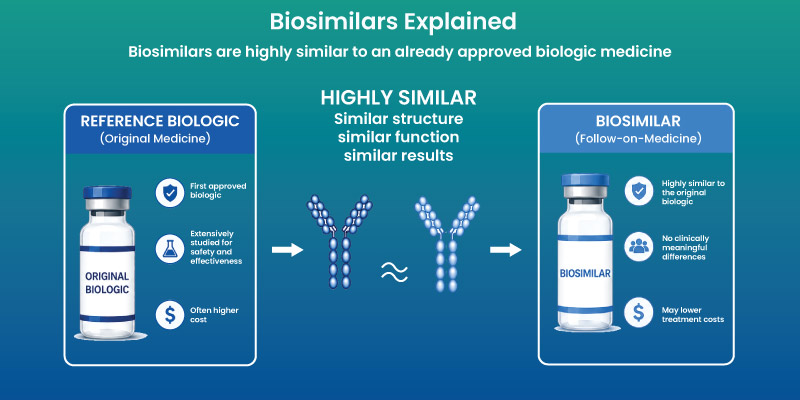
How Biosimilar Medications Work
Role of Living Cells in Drug Production: They are produced using living systems such as bacteria or mammalian cells. This process introduces variability, requiring strict quality control.
Why Biosimilars Are Not Identical: Minor differences may exist due to manufacturing processes. These differences do not impact clinical performance when regulatory criteria are met.
They function through the same biological pathways as their reference products, targeting specific proteins or immune responses.
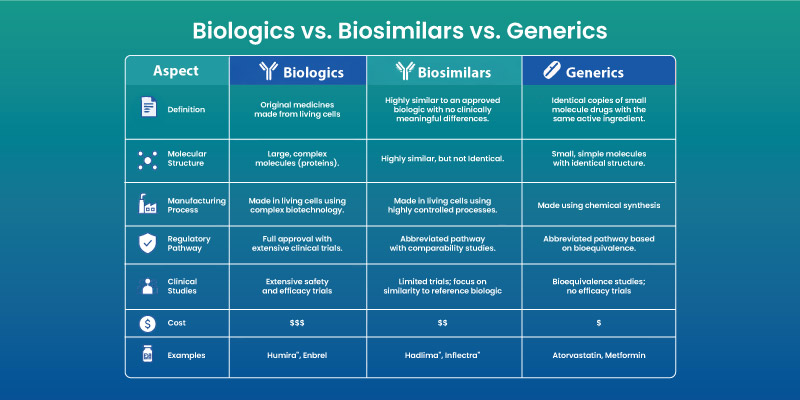
Biosimilars vs Biologics vs Generics
Key Differences Table
| Feature | Biologics | Biosimilars | Generics |
| Source | Living cells | Living cells | Chemical synthesis |
| Replication | Original | Highly similar | Identical |
| Complexity | High | High | Low |
| Cost | High | Lower | Lowest |
Why Biosimilars Are Not Generics: Generics replicate small-molecule drugs exactly. They cannot achieve exact duplication due to biological complexity.
Clinical Equivalence: Regulatory approval requires demonstration of no meaningful differences in safety or effectiveness compared to the reference biologic.
Examples of Biosimilar Drugs
Several of them approved for clinical use across therapeutic areas:
- Infliximab biosimilars for autoimmune conditions
- Trastuzumab biosimilars in oncology
- Adalimumab biosimilars for inflammatory diseases
These examples demonstrate expanding adoption in high-cost treatment areas.
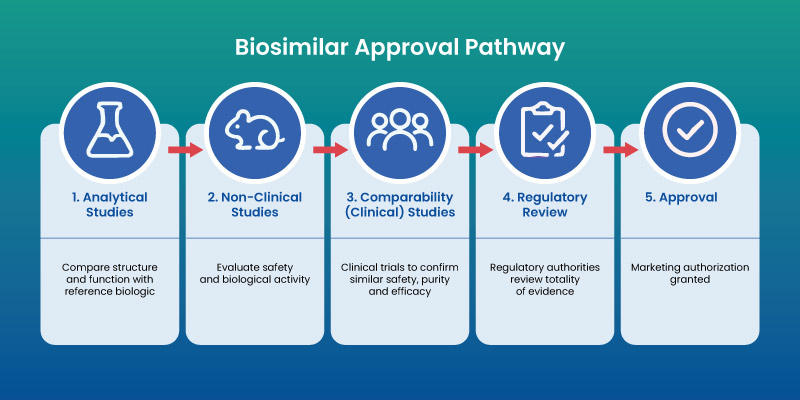
FDA and Biosimilars: Approval Process
Approval involves a stepwise evaluation:
- Analytical comparison with reference product
- Non-clinical studies
- Clinical trials assessing safety and efficacy
- Post-marketing surveillance
Regulatory agencies require robust evidence to ensure consistency and patient safety.
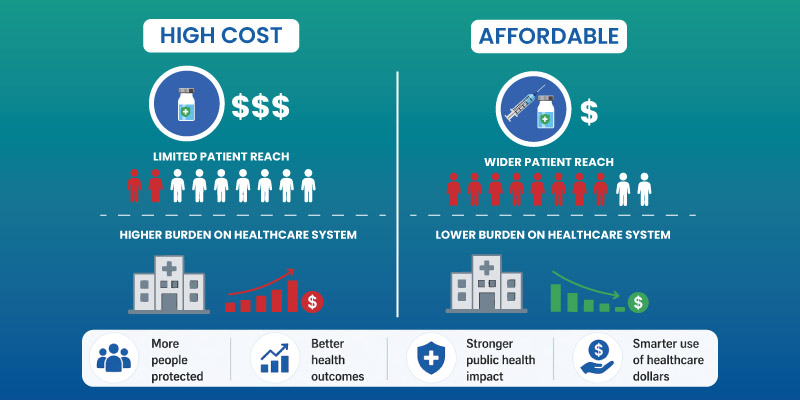
Benefits of Biosimilar Pharmaceuticals
They offer measurable advantages:
- Reduced treatment costs
- Increased patient access to advanced therapies
- Competitive pricing within healthcare systems
- Support for long-term treatment sustainability
These benefits are particularly relevant in chronic disease management.
Uses of Biosimilars in Medicine
They are used across multiple therapeutic areas:
- Oncology
- Rheumatology
- Gastroenterology
- Endocrinology
Their role continues to expand as more products receive approval.
Safety, Risks & Effectiveness
Clinical evidence supports comparable safety and effectiveness. However, monitoring remains essential.
Key considerations include:
- Immunogenic response variability
- Long-term safety tracking
- Physician and patient confidence
Regulatory oversight ensures ongoing evaluation after approval.
Biosimilars in Pharmacology
From a pharmacological perspective, they follow the same therapeutic pathways as biologics. Pharmacokinetics and pharmacodynamics are evaluated to confirm comparable absorption, distribution, and activity.
This ensures predictable clinical performance across patient populations.
Cost of Biosimilar Drugs
Cost efficiency is a primary driver of biosimilar adoption globally.
| Factor | Impact |
| Development cost | Lower than biologics |
| Market competition | Drives price reduction |
| Healthcare access | Expands treatment availability |
Cost efficiency is a primary driver of biosimilar adoption globally.
Challenges in Biosimilar Adoption
Adoption is influenced by several barriers:
- Limited awareness among patients
- Physician hesitation in switching treatments
- Regulatory differences across regions
- Manufacturing complexity
Addressing these challenges requires education and policy alignment.
Future of Biosimilars
This market is expected to expand as patents for biologics expire. Growth will be driven by:
- Increased regulatory approvals
- Technological advancements in manufacturing
- Greater acceptance among healthcare providers
- Expansion into new therapeutic areas
This trajectory positions them as a long-term component of healthcare systems.
Read also about Medication Errors
Conclusion
Biosimilars are transforming access to biologic therapies by balancing cost efficiency with clinical reliability. As regulatory frameworks evolve and adoption increases, they are expected to play a central role in improving healthcare accessibility and sustainability.
Explore more data-driven healthcare insights and global perspectives: MDForLives
Frequently Asked Questions
Are biosimilars safe and effective?
Yes, they are required to demonstrate comparable safety and efficacy through rigorous regulatory evaluation before approval.
Who regulates biosimilars?
Regulatory bodies such as the FDA and EMA oversee approval and monitoring processes.
Why are biosimilars cheaper than biologics?
They have lower development costs and benefit from established reference product data.
Do biosimilars have the same quality standards?
Yes, they must meet strict regulatory standards for manufacturing and consistency.
Can pharmacists substitute biosimilars?
Substitution policies vary by region and depend on regulatory designation and prescribing guidelines.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.