Erenumab is a monoclonal antibody developed for the prevention of migraine, including episodic and chronic forms. Marketed as Aimovig (erenumab), it targets the calcitonin gene-related peptide receptor pathway implicated in migraine pathophysiology. Clinical trials have demonstrated significant reductions in monthly migraine days, improved quality-of-life measures, and sustained efficacy over long-term follow-up. This article reviews its uses, dosing, mechanism of action, safety profile, and emerging evidence.
Erenumab Uses
It is approved for:
- Prevention of episodic migraine
- Prevention of chronic migraine
- Reduction in monthly migraine days
- Decreasing reliance on acute rescue medications
It is not indicated for acute migraine treatment. It is a preventive biologic therapy.
Erenumab Dosage & Administration
Erenumab injection is administered subcutaneously using an autoinjector pen.
Standard Dosing
- 70 mg once monthly
- 140 mg once monthly, depending on response
Its dosing may be adjusted based on therapeutic response and tolerability.
There is no tablet formulation. The drug is available only as a subcutaneous injection.
Its administration is designed for self-injection after clinical instruction.
How Erenumab Works?
Development, Pharmacokinetics and Pharmacodynamics of Erenumab
It is a fully human monoclonal antibody targeting the CGRP receptor. After subcutaneous injection:
- Peak plasma concentration occurs in approximately 4 to 6 days
- Effective half-life is approximately 28 days
- Monthly administration maintains steady-state levels
Pharmacodynamically, it blocks receptor activation without crossing the blood-brain barrier extensively.
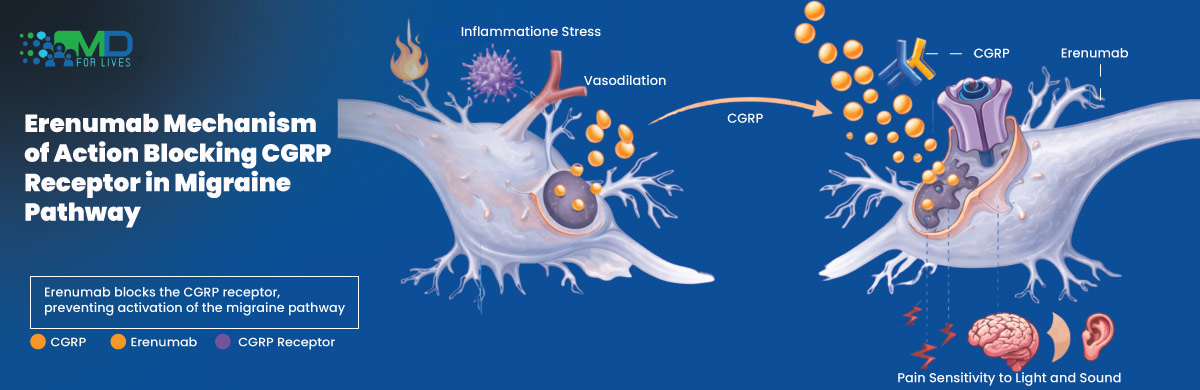
Erenumab Mechanism of Action
Migraine pathophysiology involves activation of the trigeminovascular system and release of CGRP, a potent vasodilator and pain mediator.
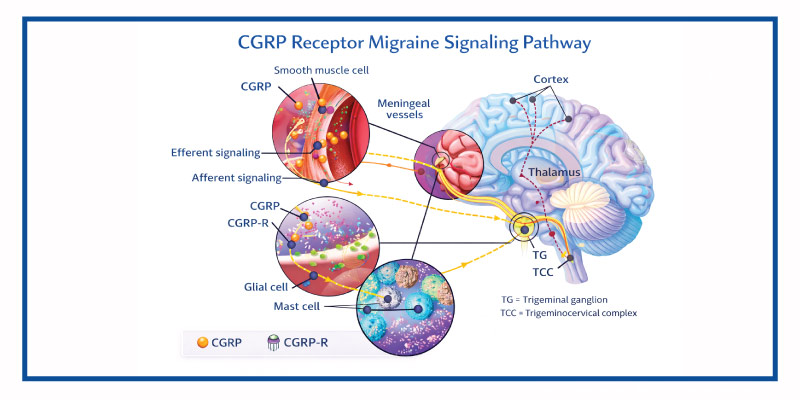
Its mechanism of action involves:
- Binding to the CGRP receptor
- Preventing CGRP from activating downstream inflammatory and nociceptive pathways
- Reducing neurogenic inflammation
- Decreasing migraine frequency
Unlike small-molecule CGRP antagonists, it specifically targets the receptor rather than the peptide.
Role of Erenumab in the Treatment of Migraine
Migraine remains a leading cause of disability globally. Traditional preventive treatments include beta-blockers, anticonvulsants, and antidepressants, many with systemic adverse effects.
It is particularly useful in:
- Patients with inadequate response to oral preventives
- Chronic migraine cases
- Patients intolerant to conventional therapies
Erenumab: Clinical Data in Migraine
Multiple phase II and III erenumab clinical trials evaluated efficacy in episodic and chronic migraine populations.
Key findings include:
- Reduction of 3 to 4 monthly migraine days in episodic migraine
- Reduction of 6 to 7 monthly migraine days in chronic migraine
- Sustained response in long-term extension studies
- Improvement in patient-reported disability scales
Migraine-Related Disability and Quality of Life Scales in Erenumab Trials
Clinical trials utilized:
- MIDAS (Migraine Disability Assessment)
- HIT-6 (Headache Impact Test)
- MSQ (Migraine-Specific Quality of Life Questionnaire)
Results showed statistically significant improvements compared to placebo groups.
Common Side Effects of Erenumab
Reported adverse effects include:
- Injection site reactions
- Constipation
- Muscle cramps
- Rare hypersensitivity reactions
Drug–Drug Interactions
It has minimal known drug–drug interactions due to its monoclonal antibody structure and lack of hepatic metabolism via cytochrome P450 pathways.
Safety and Tolerability of Erenumab
Long-term safety data show:
- Stable adverse event rates over time
- Low discontinuation rates
- Minimal cardiovascular safety signals
However, post-marketing surveillance continues to evaluate rare events.
Limitations in the Use of Erenumab
Despite clinical benefits, limitations include:
- Cost considerations
- Insurance coverage variability
- Injection route preference barriers
- Limited long-term data beyond five years
Erenumab: Future Directions
Emerging research is exploring:
- Combination therapy strategies
- Predictive biomarkers for response
- Real-world effectiveness data
- Long-term cardiovascular outcomes
List of Studies & Clinical Trials So Far
Major trials include:
- ARISE trial
- STRIVE trial
- LIBERTY trial
- Long-term open-label extension studies
These collectively demonstrate significant migraine day reduction and sustained tolerability.
Conclusions
Erenumab represents a targeted, receptor-specific preventive therapy for migraine. By inhibiting the CGRP receptor pathway, it reduces migraine frequency and disability with a favorable tolerability profile. As clinical data accumulates, it continues to shape modern migraine prevention strategies.
Join a global healthcare intelligence community shaping neurology insights. Explore MDForLives
Frequently Asked Questions
What is the success rate of erenumab?
Clinical trials report that approximately 40 to 50 percent of patients achieve at least a 50 percent reduction in monthly migraine days, depending on migraine subtype.
How long does it take for erenumab to work?
Some patients report improvement within the first month. Full therapeutic evaluation is typically assessed after three months.
How long does erenumab last?
Each injection lasts approximately one month. The half-life supports monthly dosing.
Does erenumab cause hair loss?
Hair loss is not a commonly reported adverse effect in major clinical trials. Post-marketing data continue to monitor rare events.
What is the best medication for chronic migraines?
Treatment depends on patient profile. Options include CGRP inhibitors such as erenumab, onabotulinumtoxinA, and traditional oral preventives. Clinical evaluation is required.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.