NTRK fusion is a rare genetic alteration that can drive cancer growth across multiple tumor types. It is not defined by where the cancer starts but by a specific molecular change that keeps cell growth signals permanently active. This makes it clinically important despite its low frequency. With the development of targeted therapies, identifying this fusion now directly influences treatment decisions and outcomes.
What Is NTRK Fusion?
NTRK fusion occurs when one of the NTRK genes joins with another gene, forming a hybrid gene that produces an abnormal protein. This protein sends continuous signals for cell division, bypassing normal control mechanisms.
There are three NTRK genes. Fusions involving any of them can lead to cancer. The key point is not the gene itself, but the persistent activation of growth signaling that follows.
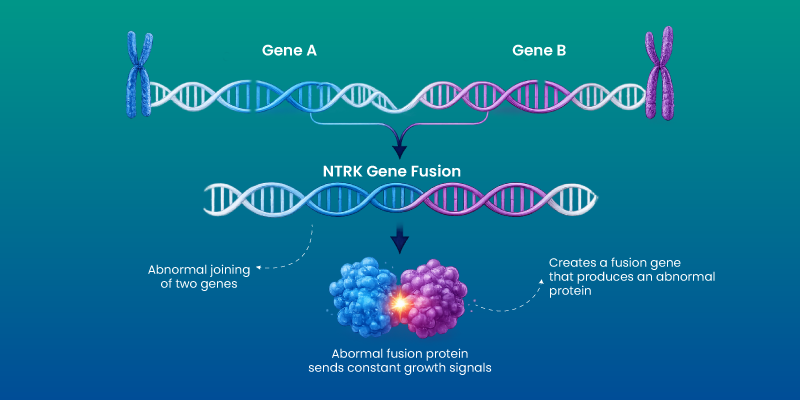
How NTRK Fusion Causes Cancer
Under normal conditions, TRK proteins respond only when triggered by specific biological signals. In fusion-driven tumors, this control is lost.
The fusion protein remains active without regulation. This leads to:
- Continuous cell proliferation
- Resistance to programmed cell death
- Increased tumor survival
Because the mechanism is specific, blocking this signal can stop tumor growth in a targeted way.
Types of Cancers with NTRK Fusion
This alteration appears in both rare and common cancers.
It is frequently found in certain rare tumors, including infantile fibrosarcoma and some salivary gland cancers. In more common cancers such as lung or thyroid cancer, it appears in a small percentage of cases.
This distribution makes testing important. A rare mutation in a common cancer can still define treatment.
How Common Is NTRK Fusion?
NTRK fusion is uncommon in most cancers, typically occurring in less than one percent of cases. However, in selected rare tumors, it can be present in a majority of patients.
Its clinical value does not depend on prevalence. Instead, it lies in its predictability as a treatment target. When present, it creates a clear pathway for therapy selection.
Symptoms and Clinical Presentation
There are no symptoms unique to this fusion. Clinical presentation depends entirely on where the tumor is located.
A tumor in the lung may cause cough or breathlessness. A brain tumor may lead to neurological symptoms. Soft tissue tumors may present as localized swelling.
Because symptoms are nonspecific, molecular testing becomes essential for detection.
Diagnosis and Testing for NTRK Fusion
Detection requires genomic analysis rather than routine imaging.
| Method | Use |
| Next-generation sequencing | Comprehensive detection of gene fusions |
| Immunohistochemistry | Screening tool |
| FISH testing | Confirms gene rearrangement |
Testing is usually done in advanced disease or when considering targeted therapy. In some settings, broader genomic profiling includes NTRK as part of a panel.

Treatment Options for NTRK Fusion Cancer
The introduction of TRK inhibitors has changed treatment strategy.
These drugs block the abnormal signaling caused by the fusion protein. Unlike chemotherapy, which targets rapidly dividing cells broadly, these therapies act on a specific molecular pathway.
Clinical data shows high response rates across different tumor types. Patients often experience rapid tumor shrinkage after treatment initiation.
Second-generation inhibitors are being developed to address resistance, which can emerge over time.

Prognosis and Outcomes
Outcomes vary depending on tumor type, stage, and access to treatment.
Patients receiving targeted therapy often show meaningful responses, including symptom relief and tumor reduction. In some cases, disease control is sustained for extended periods.
However, resistance remains a challenge. Tumors may adapt, requiring changes in treatment strategy.
NTRK Fusion in Precision Oncology
NTRK fusion is one of the clearest examples of tumor-agnostic treatment.
Traditional oncology classifies cancer by organ. Precision oncology focuses on genetic drivers instead. This shift allows the same drug to be used across different cancers if they share the same mutation.
This model improves treatment alignment and avoids unnecessary therapies that do not target the underlying mechanism.

NTRK Fusions and Emerging Therapeutics
Current research is focused on improving durability of response.
Areas of development include:
- Next-generation inhibitors targeting resistance mutations
- Combination therapies to extend response duration
- Integration with broader genomic treatment strategies
These approaches aim to move from short-term response to sustained disease control.
Read also about Gene Therapy
Conclusion
NTRK fusion represents a small but critical subset of cancer biology. Its significance lies in its direct link to targeted therapy. Identifying this alteration changes treatment pathways, often leading to better outcomes compared to conventional approaches. As testing becomes more accessible, its role in clinical decision-making will continue to expand.
Explore more evidence-led healthcare insights: MDForLives
Frequently Asked Questions
Is NTRK fusion hereditary?
No. It occurs in tumor cells and is not inherited.
Should all cancer patients be tested for NTRK fusion?
Testing is considered in advanced disease or when treatment options are limited.
When should NTRK testing be done?
Typically during molecular profiling or when standard treatments fail.
What is the survival rate for NTRK fusion cancer?
It varies widely depending on tumor type and treatment response.
Is NTRK fusion cancer aggressive?
Aggressiveness depends on the tumor itself, not the fusion alone.
Can NTRK fusion cancers be cured?
Some cases respond well to treatment, but long-term outcomes vary.

MDForLives is a global healthcare intelligence platform where real-world perspectives are transformed into validated insights. We bring together diverse healthcare experiences to discover, share, and shape the future of healthcare through data-backed understanding.